
Carbon brushes for automobile electrical equipment
For driving various motors for electric power steering systems, fuel injection pumps, ABS, windshield wipers, power windows, throttle control, power door locks and power door mirrors,etc.

Carbon brushes for electric power tools
For driving various motors for circular saws, electric drills,grinders,impact drivers and electric drivers,etc.

Carbon brushes for vacuum cleaners
For driving various motors for household-use vacuum cleaners.

Carbon brushes for micro motors
For driving various motors for hair driers, electric toothbrushes, cameras, personal computers, printers and automatic vending machines.
Carbon brushes for slip-rings
Ferris wheels, aerogenerators, and auto reels, etc.


Aupac carbon brushes are made using a process known as "powder metallurgical technology." Producing metal products and materials using powder metallurgical technology involves mixing and blending several types of metal powder at a proper ratio, placing it in a mold, compacting and pressing it at high pressures, and then putting it in an oven for sintering at a temperature below the metal-fusion point. The term "powder metallurgical technology" is also often used to describe processes involving ingredients other than metal powder, such as carbon and oxide powder.










Substances composed of the element carbon include diamond, graphite, coke and the carbon nanotube which is attracting much attention recently. Among such various allotropes of carbon, graphite in particular has some notable properties, such as:
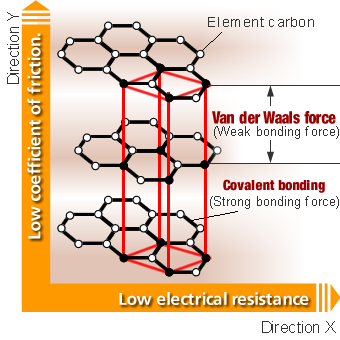
Low coefficient of friction
High chemical resistance
High heat resistance
Such properties arise from the graphite's characteristic crystal structure.
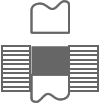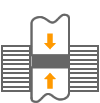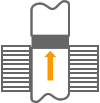
Graphite has a layered crystal structure; within each layer (direction X), the carbon atoms are held together in a strong covalent bond.
On the other hand, the atoms in different layers (direction Y) are held together in a weak van der Waals force.
Because of such crystal structure, the amount of electrical resistance within each layer (direction X) of graphite is similar to metals, while the bonding force between each layer (direction Y) is very weak. Because of this property, layers of graphite easily separate when an outside force is applied, which translates into a low coefficient of friction.
In our daily lives, graphite is found inside pencils: writing is possible because graphite layers separate and attach onto paper; because each graphite layer separates without much friction, papers are not torn apart.
Carbon brushes need to be built of materials which can receive and supply large amounts of electric current to and from objects rotating (moving) at high speeds using low friction force, and which can endure heating and oxidation. Graphite, with material properties ideally matched to such demands, is the natural choice for carbon brushes.
Production of graphite involves heat decomposition of organic substances under airtight conditions to obtain a substance with high carbon content. The process is called carbonization and the derived substance is called, carbon. Then the substance is further processed by subjecting it to high temperatures (over 2,500 degrees Celsius), also under airtight conditions, to develop the crystal structure, and the result of this process known as graphitization, is graphite. Such graphite obtained through artificial means is known as artificial graphite, whereas graphite formed naturally is called natural graphite.
There is a wide variety of substances composed of the element carbon; it is a special element constituting an important part of every organic matter and playing a vital role for life on earth. Because of such special qualities, carbon is often referred to as “black art.” News of the carbon nanotube along with other carbon-related discoveries in recent years have further confirmed such magical powers of this very special element.